Benefits of Eyebright Extract Powder
2026-04-02 14:13:48
Eyebright extract powder offers remarkable therapeutic potential for modern nutraceutical and pharmaceutical applications, delivering standardized bioactive compounds including flavonoids, iridoid glycosides, and tannins. Derived from Euphrasia officinalis, this greenish-brown to brown powder provides exceptional anti-inflammatory, astringent, and antioxidant properties that support ocular health and address digital eye strain. With centuries of traditional European usage backing its efficacy, this premium botanical ingredient serves diverse applications from dietary supplements and functional beverages to cosmeceutical formulations, making it an invaluable asset for manufacturers seeking evidence-based natural solutions.
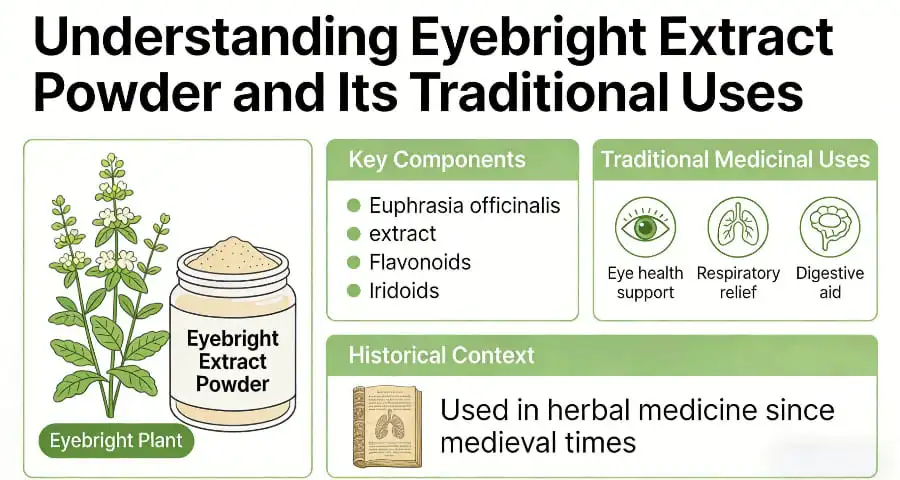
Understanding Eyebright Extract Powder and Its Traditional Uses
Eyebright extract powder originates from the aerial parts of Euphrasia officinalis, a member of the Scrophulariaceae family that has garnered respect in European traditional medicine for over six centuries. The 14th-century European Pharmacopoeia documented its remarkable efficacy against various ocular ailments, establishing a foundation of therapeutic credibility that continues to influence modern applications.
Historical Significance and Traditional Applications
The herb's reputation as a vision-supporting botanical stems from its comprehensive phytochemical profile, particularly its concentration of aucubin, quercetin, apigenin, and astringent tannins. Traditional practitioners utilized eyebright preparations to address conjunctival irritation, seasonal eye allergies, and general ocular discomfort through teas, tinctures, and topical washes.
Active Compound Profile and Therapeutic Mechanisms
Modern analytical methods reveal that eyebright extract contains approximately 2-4% flavonoids and 0.5-1% iridoid glycosides, with aucubin serving as the primary biomarker for standardization. These compounds work synergistically to provide anti-inflammatory effects through COX-2 pathway inhibition while delivering potent antioxidant protection via free radical scavenging mechanisms.
Procurement professionals benefit from understanding these traditional foundations when evaluating supplier credentials and product specifications. The transition from folk medicine to standardized extracts requires rigorous attention to extraction ratios, typically ranging from 4:1 to 10:1, ensuring consistent bioactive delivery across manufacturing batches.
Key Benefits of Eyebright Extract Powder for Eye Health and Wellness
The therapeutic advantages of eyebright extract powder extend beyond traditional applications, addressing contemporary health challenges while providing manufacturers with versatile formulation opportunities. Clinical observations and phytochemical research demonstrate multiple mechanisms supporting ocular and systemic wellness.
Anti-Inflammatory and Antioxidant Properties
Eyebright's flavonoid content delivers significant anti-inflammatory benefits, particularly valuable for addressing digital eye strain and environmental oxidative stress. The extract's ability to reduce ocular inflammation surpasses many synthetic alternatives through natural pathways that support tissue healing without adverse effects commonly associated with artificial solutions.
Research indicates that the extract's ORAC (Oxygen Radical Absorbance Capacity) values demonstrate superior antioxidant potential compared to conventional eye care ingredients. This characteristic proves especially valuable for protecting delicate eye tissues against blue light exposure and environmental pollutants that increasingly affect modern consumers.
Enhanced Immune Defense and Allergy Relief
The immunomodulatory properties of euphrasia extract provide substantial advantages for individuals experiencing seasonal allergies and environmental sensitivities. Unlike single-action alternatives such as goldenseal, eyebright offers comprehensive support through multiple therapeutic pathways, including histamine response modulation and mucosal membrane strengthening.
Systemic Wellness Benefits Beyond Ocular Health
Beyond eye-specific applications, the extract contributes to overall wellness through its digestive support properties and general anti-inflammatory effects. This multifunctional profile creates opportunities for manufacturers developing comprehensive wellness formulations that address multiple health concerns within single products.
The astringent properties derived from tannin content provide additional therapeutic value, particularly in topical applications where tissue tightening and pore refinement benefit cosmetic formulations. These diverse benefits position eyebright extract as a strategic ingredient for companies seeking to differentiate their products in competitive markets.
Choosing the Right Eyebright Extract Powder for Your Business Needs
Selecting appropriate eyebright extract powder requires careful consideration of source materials, processing methods, and quality specifications that align with intended applications. The decision between organic and conventional sources significantly impacts both product positioning and manufacturing requirements.
Organic versus Conventional Sourcing Considerations
Organic eyebright extract typically commands premium pricing while offering enhanced marketing appeal and potentially superior bioactive profiles. Organic certification ensures cultivation without synthetic pesticides or fertilizers, resulting in cleaner extraction profiles that benefit sensitive applications such as eye drops or cosmetic formulations.
Conventional sources may provide cost advantages for large-volume applications while maintaining therapeutic efficacy when sourced from reputable suppliers with stringent quality controls. The choice depends on target market expectations, regulatory requirements, and budget considerations that vary across different product categories.
Form Factor Selection and Manufacturing Compatibility
The selection between powder, capsule-ready granules, or liquid-soluble grades directly influences manufacturing workflows and final product characteristics. Standard powder forms suit tablet and capsule production but may require additional processing for clear liquid applications.
Here are the critical factors influencing form factor selection:
- Particle size distribution: 95% passage through 80 mesh ensures optimal blending homogeneity for solid dosage forms
- Bulk density specifications: Critical for accurate dosing in automated encapsulation equipment
- Solubility characteristics: Water-soluble grades prevent sedimentation in functional beverages
- Moisture content: Typically maintained below 5% to prevent degradation during storage
These technical specifications require close collaboration between procurement teams and suppliers to ensure compatibility with existing manufacturing infrastructure while maintaining product quality standards.
Supplier Qualification and Certification Requirements
Due diligence in supplier selection involves verifying GMP compliance, ISO certifications, and analytical testing capabilities that ensure consistent product quality. Established suppliers demonstrate their commitment through comprehensive documentation, including Certificates of Analysis, stability studies, and regulatory compliance records.
Quality assurance extends beyond basic certifications to encompass supply chain transparency, raw material traceability, and contamination control measures that protect against heavy metals and microbial contamination. These factors become particularly critical for pharmaceutical and cosmetic applications where regulatory scrutiny demands exceptional purity standards.
Evaluating Eyebright Extract Powder in Comparison with Alternative Solutions
Market analysis reveals eyebright's competitive advantages when compared to alternative eye health ingredients, positioning it uniquely within the natural products landscape. Understanding these comparative benefits helps manufacturers make informed decisions regarding product formulation and positioning strategies.
Comparative Analysis with Competing Ingredients
Euphrasia officinalis extract offers distinct advantages over traditional alternatives such as chamomile, bilberry, and lutein through its combination of anti-inflammatory, astringent, and immune-supporting properties. While chamomile provides gentle anti-inflammatory effects, it lacks the specific ocular targeting that eyebright delivers through its specialized phytochemical profile.
Bilberry extract, though popular for vision support, primarily addresses circulation and antioxidant needs without providing the comprehensive inflammatory response modulation that eyebright offers. Lutein focuses specifically on macular protection but lacks the broader therapeutic scope that makes eyebright suitable for multiple application areas.
Market Dynamics and Consumer Preferences
Growing consumer awareness of digital eye strain has increased demand for natural solutions that address modern lifestyle challenges. Market research indicates a 15-20% annual growth rate in eye health supplement categories, with natural ingredients showing particular strength among health-conscious demographics.
The trend toward clean-label formulations favors botanical extracts over synthetic alternatives, creating opportunities for manufacturers who can effectively communicate eyebright's traditional heritage combined with modern quality standards. Consumer education about eyebright's European medicinal history provides compelling marketing narratives that resonate with target audiences seeking authentic natural solutions.
Regulatory Compliance and Safety Considerations
FDA and EMA guidelines for botanical ingredients require careful attention to standardization, labeling accuracy, and contraindication disclosure. Eyebright extract maintains an excellent safety profile when properly standardized and used within recommended dosage ranges, typically equivalent to 2-4 grams of dried herb daily.
Regulatory compliance demands transparent communication regarding potential interactions, particularly the tannin content's ability to precipitate proteins or alkaloids in complex formulations. Stability testing becomes essential when combining eyebright with other active ingredients to prevent precipitation or potency loss during product shelf life.
Optimizing Procurement and Usage of Eyebright Extract Powder
Successful integration of eyebright extract into product lines requires strategic procurement planning, proper storage protocols, and comprehensive supplier relationships that ensure consistent quality and supply continuity. These operational considerations directly impact product success and customer satisfaction.
Strategic Sourcing and Supply Chain Management
Effective procurement strategies prioritize establishing relationships with suppliers who demonstrate consistent quality, competitive pricing, and reliable delivery schedules. Bulk purchasing agreements often provide cost advantages while ensuring adequate inventory for production planning and market demand fluctuations.
Supply chain optimization involves understanding seasonal availability patterns, storage requirements, and transportation considerations that affect extract stability. Eyebright extract maintains optimal potency when stored in airtight containers away from moisture and light, with proper storage extending shelf life to 24-36 months under controlled conditions.
Quality Assurance and Testing Protocols
Comprehensive quality control programs encompass incoming raw material testing, in-process monitoring, and finished product verification to ensure consistent bioactive content and purity standards. Key analytical parameters include flavonoid content verification, microbial testing, heavy metal analysis, and residual solvent detection.
Partnership with suppliers who provide detailed Certificates of Analysis, stability data, and regulatory documentation streamlines quality assurance processes while reducing internal testing burdens. Regular supplier audits and quality reviews maintain consistent standards and identify potential improvement opportunities.
Technical Support and Application Development
Leading suppliers invest in technical support services that help customers optimize formulations, resolve compatibility issues, and develop innovative applications. This collaborative approach accelerates product development timelines while minimizing formulation risks and market entry delays.
Application development support includes stability testing assistance, dosage optimization guidance, and regulatory documentation support that facilitates product registration and market approval processes. These value-added services differentiate premium suppliers from commodity providers while building long-term partnership relationships.
Conclusion
Eyebright extract powder represents a compelling opportunity for manufacturers seeking to capitalize on growing demand for natural eye health solutions. Its unique combination of anti-inflammatory, antioxidant, and astringent properties, backed by centuries of traditional use and modern scientific validation, positions it as a versatile ingredient for diverse applications ranging from dietary supplements to cosmetic formulations. The key to successful implementation lies in partnering with qualified suppliers who provide consistent quality, comprehensive technical support, and regulatory compliance assistance that ensures product success in competitive markets.
FAQ
What standardization options are available for eyebright extract powder?
Standardization typically focuses on flavonoid content ranging from 0.5% to 2% by UV spectrophotometry, or aucubin content verified through HPLC analysis. Extraction ratios of 4:1, 8:1, and 10:1 provide different concentration levels to match specific formulation requirements and dosage targets.
How does tannin content affect formulation compatibility?
Tannin levels between 5-10% can cause precipitation when combined with proteins or alkaline compounds. Stability testing becomes essential for complex formulations, and pH adjustment or chelating agents may be required to prevent interaction issues in liquid applications.
What particle size specifications ensure optimal manufacturing performance?
Standard specifications require 95% passage through 80 mesh (180 microns) for solid dosage forms, ensuring uniform blending and consistent tablet compression. Finer grades available for specialized applications such as cosmetic formulations or liquid suspensions.
Why Choose Wellgreen as Your Eyebright Extract Supplier?
Wellgreen Technology stands as your trusted eyebright extract manufacturer, offering pharmaceutical-grade botanical extracts backed by GMP-certified manufacturing and comprehensive quality assurance. Our state-of-the-art facility maintains extensive inventory of standardized eyebright extract powder with verified potency levels and complete analytical documentation. We provide flexible OEM support, custom specifications, and rapid delivery services that meet demanding production schedules while ensuring consistent product quality. Contact our technical team at wgt@allwellcn.com to discuss your specific requirements and discover how our premium eyebright extract solutions can enhance your product portfolio with reliable, cost-effective botanical ingredients.
References
European Medicines Agency. Assessment Report on Euphrasia officinalis L. and Euphrasia rostkoviana Hayne, herba. Committee on Herbal Medicinal Products, 2015.
Barnes, Joanne, et al. Herbal Medicines: A Guide for Healthcare Professionals. 4th edition, Pharmaceutical Press, 2019.
Wichtl, Max, editor. Herbal Drugs and Phytopharmaceuticals: A Handbook for Practice on a Scientific Basis. 3rd edition, CRC Press, 2004.
Blumenthal, Mark, et al. The Complete German Commission E Monographs: Therapeutic Guide to Herbal Medicines. American Botanical Council, 1998.
Chevallier, Andrew. Encyclopedia of Herbal Medicine: 550 Herbs and Remedies for Common Ailments. 3rd edition, DK Publishing, 2016.
Mills, Simon, and Kerry Bone. The Essential Guide to Herbal Safety. Churchill Livingstone, 2005.







